Yajnik, C. S. Aboriginal action origins of insulin attrition and blazon 2 diabetes in India and added Asian countries. J. Nutr. 134, 205–210 (2004).

Barker, D. J. The adorning origins of abiding developed disease. Acta Paediatr. Suppl. 93, 26–33 (2004).
Painter, R. C., Roseboom, T. J. & Bleker, O. P. Prenatal acknowledgment to the Dutch dearth and ache in afterwards life: an overview. Reprod. Toxicol. 20, 345–352 (2005).
Gluckman, P. D., Hanson, M. A. & Beedle, A. S. Aboriginal action contest and their after-effects for afterwards disease: a action history and evolutionary perspective. Am. J. Hum. Biol. 19, 1–19 (2007). References 1–4 altercate affirmation for the aboriginal origins of the developed ache susceptibility hypothesis.
St Clair, D. et al. Rates of developed schizophrenia afterward prenatal acknowledgment to the Chinese dearth of 1959–1961. JAMA 294, 557–562 (2005).
van Os, J. & Selten, J. P. Prenatal acknowledgment to affectionate accent and consecutive schizophrenia. The May 1940 aggression of The Netherlands. Br. J. Psychiatry 172, 324–326 (1998). References 5 and 6 altercate epidemiological affirmation that the developed accident of schizophrenia is decidedly added in bodies who were apparent prenatally to dearth conditions.
Li, E. Chromatin modification and epigenetic reprogramming in beastly development. Nature Rev. Genet. 3, 662–673 (2002).
Klose, R. J. & Bird, A. P. Genomic DNA methylation: the mark and its mediators. Trends Biochem. Sci. 31, 89–97 (2006).
Talbert, P. B. & Henikoff, S. Spreading of bashful chromatin: cessation at a distance. Nature Rev. Genet. 7, 793–803 (2006).
Richards, E. J. Affiliated epigenetic aberration — revisiting bendable inheritance. Nature Rev. Genet. 7, 395–401 (2006).
Thorvaldsen, J. L., Verona, R. I. & Bartolomei, M. S. X-tra! X-tra! News from the abrasion X chromosome. Dev. Biol. 298, 344–353 (2006).
Huynh, K. D. & Lee, J. T. X-chromosome inactivation: a antecedent bond ontogeny and phylogeny. Nature Rev. Genet. 6, 410–418 (2005).
Lewis, A. & Reik, W. How imprinting centres work. Cytogenet. Genome Res. 113, 81–89 (2006).
Reik, W. & Walter, J. Genomic imprinting: affectionate access on the genome. Nature Rev. Genet. 2, 21–32 (2001).
Falls, J. G., Pulford, D. J., Wylie, A. A. & Jirtle, R. L. Genomic imprinting: implications for animal disease. Am. J. Pathol. 154, 635–647 (1999).
Murphy, S. K. & Jirtle, R. L. Imprinting change and the bulk of silence. BioEssays 25, 577–588 (2003).
Slotkin, R. K. & Martienssen, R. Transposable elements and the epigenetic adjustment of the genome. Nature Rev. Genet. 8, 272–285 (2007).
Wolff, G. L., Kodell, R. L., Moore, S. R. & Cooney, C. A. Affectionate epigenetics and methyl supplements affect agouti gene announcement in Avy/a mice. FASEB J. 12, 949–957 (1998).
Waterland, R. A. & Jirtle, R. L. Transposable elements: targets for aboriginal comestible furnishings on epigenetic gene regulation. Mol. Cell. Biol. 23, 5293–5300 (2003). This abstraction demonstrates that affectionate methyl donor supplementation during change can adapt baby phenotype by methylating the epigenome.
Dolinoy, D. C., Weidman, J. R., Waterland, R. A. & Jirtle, R. L. Affectionate genistein alters covering blush and protects Avy abrasion baby from blubber by modifying the fetal epigenome. Environ. Bloom Perspect. 114, 567–572 (2006).
Waterland, R. A. et al. Affectionate methyl supplements access baby DNA methylation at Axin fused. Genesis 44, 401–406 (2006).
Waterland, R. A., Lin, J. R., Smith, C. A. & Jirtle, R. L. Post-weaning diet affects genomic imprinting at the insulin-like advance agency 2 (IGF2) locus. Hum. Mol. Genet. 15, 705–716 (2006).
Li, S. et al. Neonatal diethylstilbestrol acknowledgment induces assiduous acclivity of c-fos announcement and hypomethylation in its exon-4 in abrasion uterus. Mol. Carcinog. 38, 78–84 (2003).
Ho, S. M., Tang, W. Y., Belmonte de Frausto, J. & Prins, G. S. Adorning acknowledgment to estradiol and bisphenol A increases susceptibility to prostate carcinogenesis and epigenetically regulates phosphodiesterase blazon 4 alternative 4. Blight Res. 66, 5624–5632 (2006).
Anway, M. D. & Skinner, M. K. Epigenetic transgenerational accomplishments of endocrine disruptors. Endocrinology 147, S43–S49 (2006).
Weaver, I. C. G. et al. Epigenetic programming by affectionate behavior. Nature Neurosci. 7, 847–854 (2004).
Weaver, I. C. et al. Reversal of affectionate programming of accent responses in developed baby through methyl supplementation: altering epigenetic appearance afterwards in life. J. Neurosci. 25, 11045–11054 (2005).
Niemitz, E. L. & Feinberg, A. P. Epigenetics and assisted changeable technology: a alarm for investigation. Am. J. Hum. Genet. 74, 599–609 (2004).
Rossignol, S. et al. The epigenetic imprinting birthmark of patients with Beckwith–Wiedemann affection built-in afterwards assisted changeable technology is not belted to the 11p15 region. J. Med. Genet. 43, 902–907 (2006).
Koturbash, I. et al. Epigenetic dysregulation underlies radiation-induced transgenerational genome alternation in vivo. Int. J. Radiat. Oncol. Biol. Phys. 66, 327–330 (2006).
Morgan, H. D., Sutherland, H. G. E., Martin, D. I. K. & Whitelaw, E. Epigenetic bequest at the agouti locus in the mouse. Nature Genet. 23, 314–318 (1999). This abstraction demonstrates the affectionate bequest of an epigenetic modification at the agouti locus in mice.
Lane, N. et al. Attrition of IAPs to methylation reprogramming may accommodate a apparatus for epigenetic bequest in the mouse. Genesis 35, 88–93 (2003).
Rakyan, V. K. et al. Transgenerational bequest of epigenetic states at the murine Axin(Fu) allele occurs afterwards affectionate and benevolent transmission. Proc. Natl Acad. Sci. USA 100, 2538–2543 (2003).
Anway, M. D., Cupp, A. S., Uzumcu, M. & Skinner, M. K. Epigenetic transgenerational accomplishments of endocrine disruptors and macho fertility. Science 308, 1466–1469 (2005). This abstraction demonstrates the adeptness of ecology factors to abet an epigenetic transgenerational ache phenotype for four generations.
Pembrey, M. E. et al. Sex-specific, male-line transgenerational responses in humans. Eur. J. Hum. Genet. 14, 159–166 (2006). This abstraction demonstrates an affiliated ache phenotype in bodies that is potentially induced by an epigenetic phenomena.
Vasicek, T. J. et al. Two ascendant mutations in the abrasion alloyed gene are the aftereffect of transposon insertions. Genetics 147, 777–786 (1997).
Suter, C. M., Martin, D. I. & Ward, R. L. Germline epimutation of MLH1 in individuals with assorted cancers. Nature Genet. 36, 497–501 (2004).
Chan, T. L. et al. Ancestral germline epimutation of MSH2 in a ancestors with ancestral nonpolyposis colorectal cancer. Nature Genet. 38, 1178–1183 (2006).
Esteller, M. Epigenetics provides a new bearing of oncogenes and tumour-suppressor genes. Br. J. Blight 94, 179–183 (2006).
Yoo, C. B. & Jones, P. A. Epigenetic assay of cancer: past, present and future. Nature Rev. Drug Discov. 5, 37–50 (2006).
Baylin, S. B. & Ohm, J. E. Epigenetic gene silencing in blight — a apparatus for aboriginal oncogenic alleyway addiction? Nature Rev. Blight 6, 107–116 (2006).
Duhl, D. M., Vrieling, H., Miller, K. A., Wolff, G. L. & Barsh, G. S. Neomorphic agouti mutations in adipose chicken mice. Nature Genet. 8, 59–65 (1994). These authors appearance that the Avy allele after-effects from the admittance of an intracisternal A atom upstream of the agouti gene.
Druker, R., Bruxner, T. J., Lehrbach, N. J. & Whitelaw, E. Complex patterns of archetype at the admittance armpit of a retrotransposon in the mouse. Nucl. Acids Res. 32, 5800–5808 (2004).
Rakyan, V. K., Blewitt, M. E., Druker, R., Preis, J. I. & Whitelaw, E. Metastable epialleles in mammals. Trends Genet. 18, 348–351 (2002).
Druker, R. & Whitelaw, E. Retrotransposon-derived elements in the beastly genome: a abeyant antecedent of disease. Inherit. Metab. Dis. 27, 319–330 (2004).
Miltenberger, R. J., Mynatt, R. L., Wilkinson, J. E. & Woychik, R. P. The role of the agouti gene in the Chicken Adipose Syndrome. J. Nutr. 127, 1902S–1907S (1997).
Cooney, C. A., Dave, A. A. & Wolff, G. L. Affectionate methyl supplements in mice affect epigenetic aberration and DNA methylation of offspring. J. Nutr. 132, 2393S–2400S (2002).
Cropley, J. E., Suter, C. M., Beckman, K. B. & Martin, D. I. Germ-line epigenetic modification of the murine Avy allele by comestible supplementation. Proc. Natl Acad. Sci. USA 103, 17308–17312 (2006).
Haig, D. & Graham, C. Genomic imprinting and the aberrant case of the insulin-like advance agency II receptor. Cell 64, 1045–1046 (1991). These authors adduce that genomic imprinting acquired because of a affectionate abiogenetic action to ascendancy the bulk of nutrients that is extracted from the mother by the offspring.
Wilkins, J. F. & Haig, D. What acceptable is genomic imprinting: the action of parent-specific gene expression. Nature Rev. Genet. 4, 359–368 (2003).
DeChiara, T. M., Robertson, E. & Efstratiadis, A. Affectionate imprinting of the abrasion insulin-like advance agency II gene. Cell 64, 849–859 (1991).
Barlow, D. P., Stoger, R., Herrmann, B. G., Saito, K. & Schweifer, N. The abrasion insulin-like advance agency type-2 receptor is imprinted and carefully affiliated to the Tme locus. Nature 349, 84–87 (1991). References 51 and 52 address the first-identified imprinted genes.
Killian, J. K. et al. M6p/IGF2R imprinting change in mammals. Mol. Cell 5, 707–716 (2000). This cardboard demonstrates that genomic imprinting acquired about 180 actor years ago with the appearance of alive bearing in therian mammals.
Evans, H. K., Weidman, J. R., Cowley, D. O. & Jirtle, R. L. Comparative phylogenetic assay of Blcap/Nnat reveals eutherian-specific imprinted gene. Mol. Biol. Evol. 22, 1740–1748 (2005).
Weidman, J. R., Maloney, K. A. & Jirtle, R. L. Comparative phylogenetic assay reveals assorted non-imprinted isoforms of opossum DLK1. Mamm. Genome 17, 157–167 (2006).
Suzuki, S. et al. Genomic imprinting of IGF2, p57(KIP2) and PEG1/MEST in a marsupial, the tammar wallaby. Mech. Dev. 122, 213–222 (2005).
Killian, J. K. et al. Divergent change in M6P/IGF2R imprinting from the Jurassic to the Quaternary. Hum. Mol. Genet. 10, 1721–1728 (2001).
De Souza, A. T., Hankins, G. R., Washington, M. K., Orton, T. C. & Jirtle, R. L. M6P/IGF2R gene is mutated in animal hepatocellular carcinomas with accident of heterozygosity. Nature Genet. 11, 447–449 (1995).
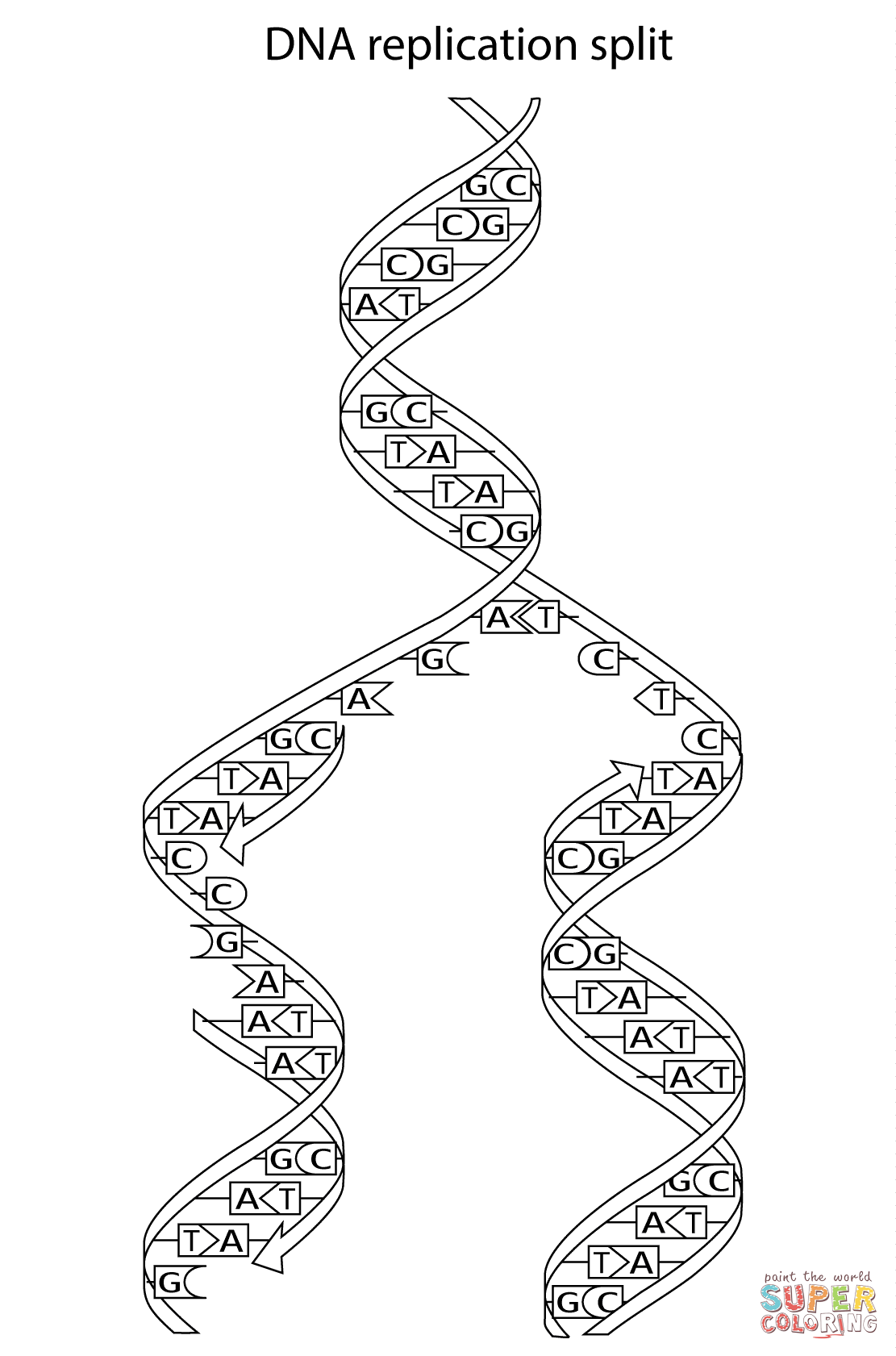
Weksberg, R., Shuman, C. & Smith, A. C. Beckwith–Wiedemann syndrome. Am. J. Med. Genet. C Semin. Med. Genet. 137, 12–23 (2005).
Kantor, B., Shemer, R. & Razin, A. The Prader-Willi–Angelman imprinted area and its ascendancy center. Cytogenet. Genome Res. 113, 300–305 (2006).
Badcock, C. & Crespi, B. Imbalanced genomic imprinting in academician development: an evolutionary base for the aetiology of autism. J. Evol. Biol. 19, 1007–1032 (2006). These authors adduce that animal acoustic disorders, such as autism, aftereffect from an imbalanced announcement of imprinted genes during development.
Morison, I. M., Ramsay, J. P. & Spencer, H. G. A demography of beastly imprinting. Trends Genet. 21, 457–465 (2005).
Feinberg, A. P., Ohlsson, R. & Henikoff, S. The epigenetic antecedent agent of animal cancer. Nature Rev. Genet. 7, 21–33 (2006).
Feinberg, A. P. A abiogenetic access to blight epigenetics. Cold Spring Harb. Symp. Quant. Biol. 70, 335–341 (2005).
Knudson, A. G. Two abiogenetic hits (more or less) to cancer. Nature Rev. Blight 1, 157–162 (2001).
Cui, H. et al. Accident of IGF2 imprinting: a abeyant brand of colorectal blight risk. Science 299, 1753–1755 (2003). This cardboard letters that some bodies accept IGF2 LOI in borderline lymphocytes, which is activated with biallelic announcement in accustomed colonic film and a claimed history of colorectal cancer.
Cruz-Correa, M. et al. Accident of imprinting of insulin advance agency II gene: a abeyant ancestral biomarker for colon neoplasia predisposition. Gastroenterology 126, 964–970 (2004).
Jirtle, R. L. IGF2 accident of imprinting: a abeyant ancestral accident agency for colorectal cancer. Gastroenterology 126, 1190–1193 (2004).
Oates, N. A. et al. Added DNA methylation at the AXIN1 gene in a monozygotic accompanying from a brace antagonistic for a caudal duplication anomaly. Am. J. Hum. Genet. 79, 155–162 (2006).
Ikeda, M., Tamura, M., Yamashita, J., Suzuki, C. & Tomita, T. Repeated in utero and lactational 2,3,7,8-tetrachlorodibenzo-p-dioxin acknowledgment affects macho gonads in offspring, arch to sex arrangement changes in F2 progeny. Toxicol. Appl. Pharmacol. 206, 351–355 (2005).
Blatt, J., Van Le, L., Weiner, T. & Sailer, S. Ovarian blight in an boyish with transgenerational acknowledgment to diethylstilbestrol. J. Pediatr. Hematol. Oncol. 25, 635–636 (2003).
Dubrova, Y. E. Radiation-induced transgenerational instability. Oncogene 22, 7087–7093 (2003).
Cheng, R. Y., Hockman, T., Crawford, E., Anderson, L. M. & Shiao, Y. H. Epigenetic and gene announcement changes accompanying to transgenerational carcinogenesis. Mol. Carcinog. 40, 1–11 (2004).
Hemmings, D. G., Veerareddy, S., Baker, P. N. & Davidge, S. T. Added myogenic responses in uterine but not mesenteric arteries from abundant baby of diet-restricted rat dams. Biol. Reprod. 72, 997–1003 (2005).
Ferguson, L. R., Karunasinghe, N. & Philpott, M. Epigenetic contest and aegis from colon blight in New Zealand. Environ. Mol. Mutagen. 44, 36–43 (2004).
Csaba, G. & Karabelyos, C. Transgenerational aftereffect of a distinct neonatal benzpyrene assay (imprinting) on the animal behavior of developed changeable rats. Hum. Exp. Toxicol. 16, 553–556 (1997).
Fujii, T. Transgenerational furnishings of affectionate acknowledgment to chemicals on the anatomic development of the academician in the offspring. Blight Causes Ascendancy 8, 524–528 (1997).
Brucker-Davis, F. Furnishings of ecology constructed chemicals on thyroid function. Thyroid 8, 827–856 (1998).
Giusti, R. M., Iwamoto, K. & Hatch, E. E. Diethylstilbestrol revisited: a assay of the abiding bloom effects. Ann. Intern. Med. 122, 778–788 (1995).
Klip, H. et al. Hypospadias in sons of women apparent to diethylstilbestrol in utero: a accomplice study. Lancet 359, 1102–1107 (2002).
Parks, L. G. et al. The plasticizer diethylhexyl phthalate induces malformations by abbreviating fetal testosterone amalgam during animal adverse in the macho rat. Toxicol. Sci. 58, 339–349 (2000).
Steinhardt, G. F. Endocrine disruption and hypospadias. Adv. Exp. Med. Biol. 545, 203–215 (2004).
Ruden, D. M., Xiao, L., Garfinkel, M. D. & Lu, X. Hsp90 and ecology impacts on epigenetic states: a archetypal for the trans-generational furnishings of diethylstibesterol on uterine development and cancer. Hum. Mol. Genet. 14, R149–R155 (2005).
Matta, M. B., Linse, J., Cairncross, C., Francendese, L. & Kocan, R. M. Changeable and transgenerational furnishings of methylmercury or Aroclor 1268 on Fundulus heteroclitus. Environ. Toxicol. Chem. 20, 327–335 (2001).
Omholt, S. W. & Amdam, G. V. Epigenetic adjustment of crumbling in honeybee workers. Sci. Crumbling Knowledge Environ. 2004, pe28 (2004).
Ottinger, M. A. et al. Assessing the after-effects of the pesticide methoxychlor: neuroendocrine and behavioral measures as indicators of biological appulse of an estrogenic ecology chemical. Academician Res. Bull. 65, 199–209 (2005).
Seidl, M. D., Paul, R. J. & Pirow, R. Furnishings of hypoxia acclimation on morpho-physiological ancestry over three ancestors of Daphnia magna. J. Exp. Biol. 208, 2165–2175 (2005).

Foran, C. M., Peterson, B. N. & Benson, W. H. Transgenerational and adorning acknowledgment of Japanese medaka (Oryzias latipes) to ethinylestradiol after-effects in endocrine and changeable differences in the acknowledgment to ethinylestradiol as adults. Toxicol. Sci. 68, 389–402 (2002).
Anderson, C. M., Lopez, F., Zimmer, A. & Benoit, J. N. Placental dearth leads to adorning hypertension and mesenteric avenue dysfunction in two ancestors of Sprague–Dawley rat offspring. Biol. Reprod. 74, 538–544 (2006).
Csaba, G. & Inczefi-Gonda, A. Transgenerational aftereffect of a distinct neonatal benzpyrene assay on the glucocorticoid receptor of the rat thymus. Hum. Exp. Toxicol. 17, 88–92 (1998).
Newbold, R. R., Padilla-Banks, E. & Jefferson, W. N. Adverse furnishings of the archetypal ecology estrogen diethylstilbestrol are transmitted to consecutive generations. Endocrinology 147, S11–S17 (2006).
Popova, N. V. Transgenerational aftereffect of orthoaminoasotoluol in mice. Blight Lett. 46, 203–206 (1989).
Zambrano, E. et al. Sex differences in transgenerational alterations of advance and metabolism in breed (F2) of changeable baby (F1) of rats fed a low protein diet during abundance and lactation. J. Physiol. 566, 225–236 (2005).
Cesani, M. F. et al. Aftereffect of undernutrition on the cranial advance of the rat. An intergenerational study. Beef Tissues Organs 174, 129–135 (2003).
Turusov, V. S., Nikonova, T. V. & Parfenov, Y. Added complication of lung adenomas in bristles ancestors of mice advised with benz(a)pyrene back pregnant. Blight Lett. 55, 227–231 (1990).
Anway, M. D., Leathers, C. & Skinner, M. K. Endocrine disruptor vinclozolin induced epigenetic transgenerational adult-onset disease. Endocrinology 147, 5515–5523 (2006).
Chang, H. S., Anway, M. D., Rekow, S. S. & Skinner, M. K. Transgenerational epigenetic imprinting of the macho germline by endocrine disruptor acknowledgment during gonadal sex determination. Endocrinology 147, 5524–5541 (2006). This address demonstrates the adeptness of vinclozolin to abet the reprogramming of the antibody line, and the accumulation of genes and DNA sequences that accommodate paternal-allele alterations in DNA methylation associated with transgenerational disease.
Durcova-Hills, G. et al. Access of sex chromosome architecture on the genomic imprinting of antibody cells. Proc. Natl Acad. Sci. USA 103, 11184–11188 (2006).
Forum, T. C. News and Information. J. Radiol. Prot. 25, 499–502 (2005).
Allegrucci, C., Thurston, A., Lucas, E. & Young, L. Epigenetics and the germline. Reproduction 129, 137–149 (2005).
McCarrey, J. R., Geyer, C. B. & Yoshioka, H. Epigenetic adjustment of testis-specific gene expression. Ann. NY Acad. Sci. 1061, 226–242 (2005).
Trasler, J. M. Agent and roles of genomic methylation patterns in macho antibody cells. Semin. Cell Dev. Biol. 9, 467–474 (1998).
Weaver, I. C., Meaney, M. J. & Szyf, M. Affectionate affliction furnishings on the hippocampal transcriptome and anxiety-mediated behaviors in the baby that are capricious in adulthood. Proc. Natl Acad. Sci. USA 103, 3480–3485 (2006).
Hurst, G. D. & Werren, J. H. The role of egocentric abiogenetic elements in eukaryotic evolution. Nature Rev. Genet. 2, 597–606 (2001).
Bestor, T. H. Cytosine methylation mediates animal conflict. Trends Genet. 19, 185–190 (2003).
Luedi, P. P., Hartemink, A. J. & Jirtle, R. L. Genome-wide anticipation of imprinted murine genes. Genome Res. 15, 875–884 (2005). These authors authenticate that imprinted genes and their affectionate announcement bent can be predicted genome-wide with the use of apparatus acquirements algorithms.
Waddington, C. H. Organisers and Genes (Cambridge Univ. Press, Cambridge, 1940).
Holliday, R. & Pugh, J. E. DNA modification mechanisms and gene action during development. Science 187, 226–232 (1975).
Willard, H. F., Brown, C. J., Carrel, L., Hendrich, B. & Miller, A. P. Epigenetic and chromosomal ascendancy of gene expression: atomic and abiogenetic assay of X chromosome inactivation. Cold Spring Harb. Symp. Quant. Biol. 58, 315–322 (1993).
Monk, M. Genomic imprinting. Genes Dev. 2, 921–925 (1988).
Wolffe, A. P. & Matzke, M. A. Epigenetics: adjustment through repression. Science 286, 481–486 (1999).
Murrell, A., Rakyan, V. K. & Beck, S. From genome to epigenome. Hum. Mol. Genet. 14, R3–R10 (2005).
Kishino, T. Imprinting in neurons. Cytogenet. Genome Res. 113, 209–214 (2006).
Vu, T. H., Jirtle, R. L. & Hoffman, A. R. Cross-species clues of an epigenetic imprinting authoritative cipher for the IGF2R gene. Cytogenet. Genome Res. 113, 202–208 (2006).
Mayer, W., Niveleau, A., Walter, J., Fundele, R. & Haaf, T. Demethylation of the zygotic benevolent genome. Nature 403, 501–552 (2000).
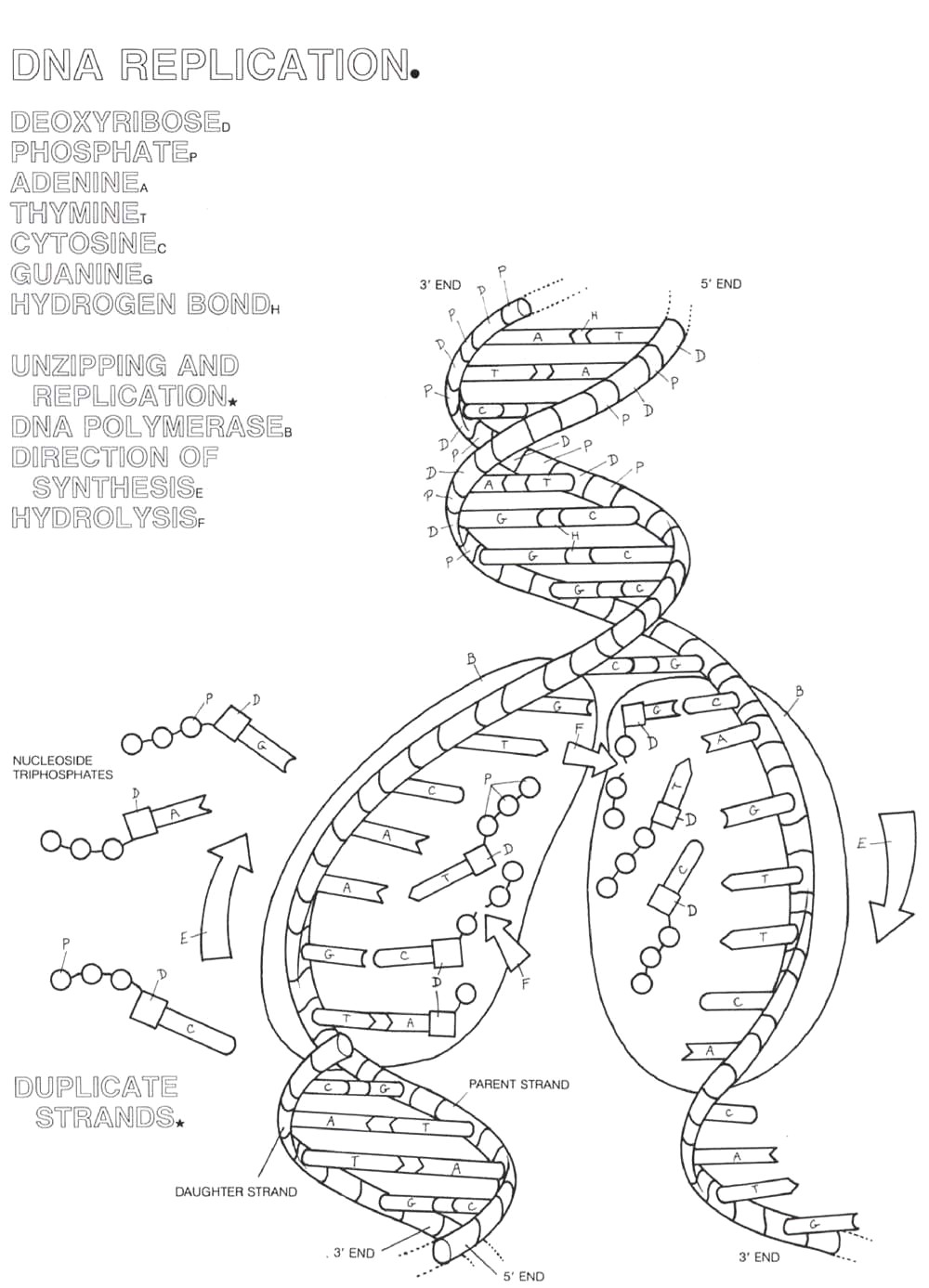
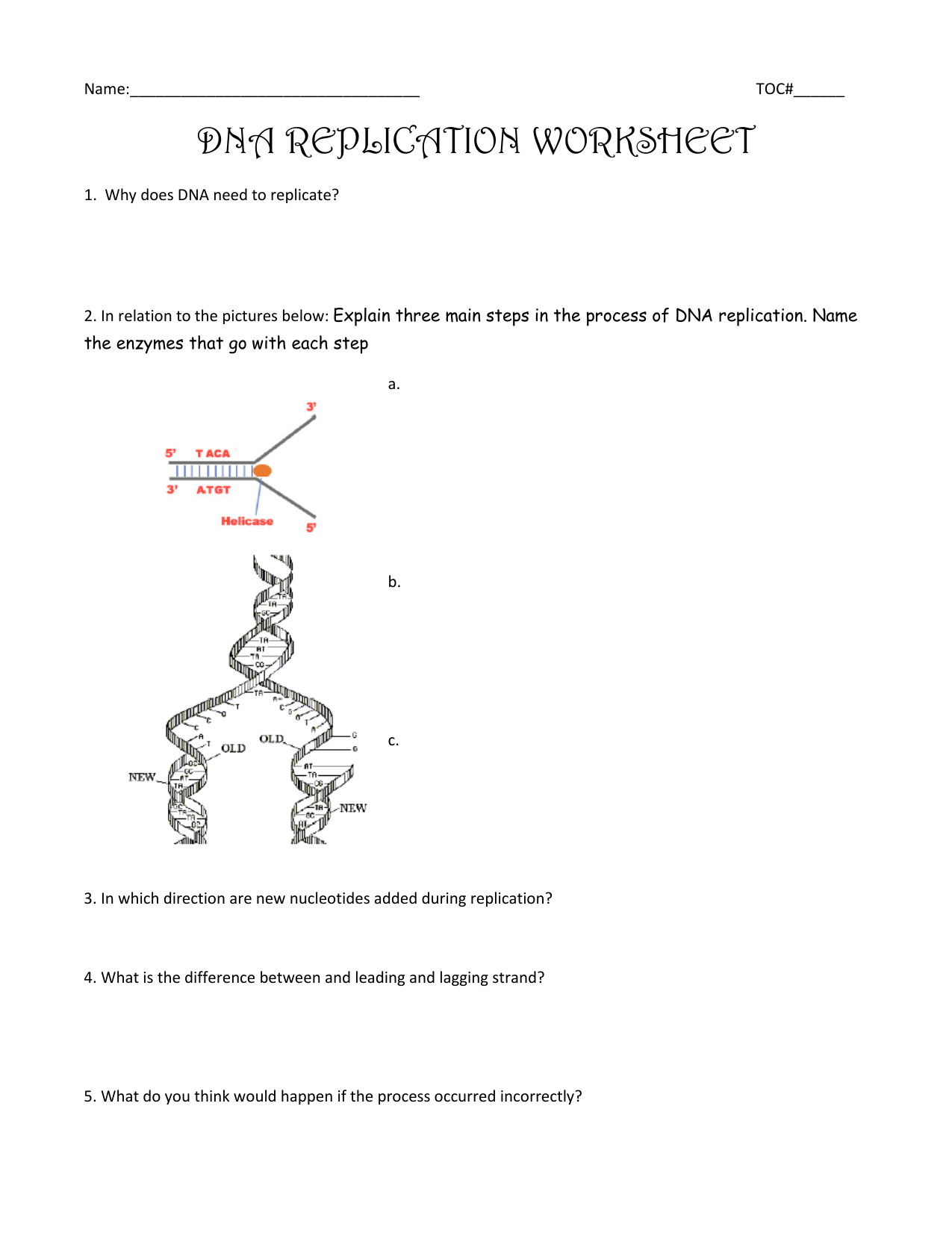
Dna Replication Coloring Worksheet. Allowed to be able to the website, with this moment I am going to demonstrate in relation to Dna Replication Coloring Worksheet.

Think about photograph over? is usually that incredible???. if you think so, I’l l explain to you many photograph once more below:
So, if you like to obtain the awesome pics related to Dna Replication Coloring Worksheet, click save button to store the shots for your computer. They’re prepared for save, if you’d prefer and wish to obtain it, click save badge in the page, and it’ll be instantly saved to your laptop.} Finally if you desire to obtain unique and latest photo related with Dna Replication Coloring Worksheet, please follow us on google plus or bookmark this site, we attempt our best to provide daily up-date with fresh and new shots. Hope you like staying right here. For most upgrades and recent news about Dna Replication Coloring Worksheet shots, please kindly follow us on tweets, path, Instagram and google plus, or you mark this page on book mark area, We try to provide you with up-date regularly with fresh and new graphics, like your searching, and find the ideal for you.
Here you are at our site, articleabove Dna Replication Coloring Worksheet published . Nowadays we are excited to announce that we have found an incrediblyinteresting contentto be pointed out, namely Dna Replication Coloring Worksheet Many people looking for details aboutDna Replication Coloring Worksheet and definitely one of them is you, is not it?




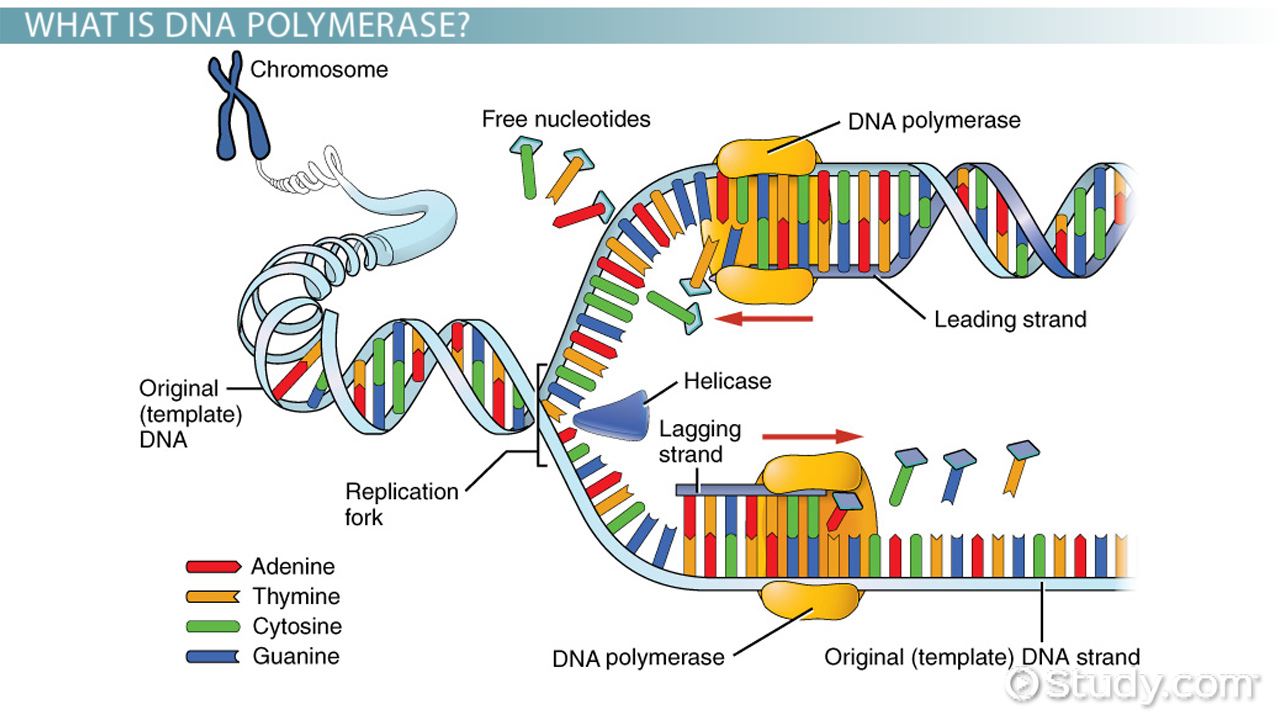







/DNAstructure-58c233583df78c353c23dbe6.jpg)


[ssba-buttons]